Independent distribution of medicines is possible because prices of individual drugs vary between EU Member States. In the Single Market, independent distributors can buy pharmaceuticals in any EU/EEA country and under strictly regulated conditions transport, repackage them to comply with national legislation and linguistic needs, and sell them at a lower price than the standard local price. This creates price competition with that same identical product sold by the original manufacturer of the medicine or its local licensee and therefore can lead to significant savings for the healthcare systems. Independent distribution of medicines also counteracts excessive pricing strategies of medicine producers and patent holders.
HOW BIG IS INDEPENDENT DISTRIBUTION?
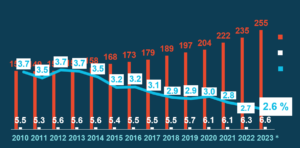
While the medicines sales in the EU have been rapidly growing over the last decade, reaching €255 billion in 2023, the turnover of EU parallel imports has remained stable in the same period at around €6 billion. In 2023, the sales of parallel imports only represented about 2.6% of the total sales, €6.6 billion, of pharmaceuticals in Europe.
The origin of the parallel imports is distributed between different European countries and, contrary to common belief, most of the parallel imported medicines are sourced in high-income countries. You can read more about it in our Trade Flow study from 2023.
The legal basis of independent distribution lays in the principles of free movement of goods and exhaustion of intellectual property (IP) rights within the Single Market. Independent distribution of medicines is not only legal but also strictly regulated under EU and national legislation.
The legality has been recognized by the Court of Justice of the European Communities since the late 1960s and 1970s. ((For patents; 29 February 1968, in Case C-24/67, Parke Davis and Co. v Probel (principle) and 31 October 1974, in case C-15/74, Centrafarm BV et Adriaan de Peijper v Sterling Drug Inc. For Trademarks; 13 July 1966, in joint cases C-56/64 and C-58/64, Établissement Consten S.à.R.L. and Grundig-Verkaufs-GmbH v Commission; 31 October 1974, in case C-16/74, Centrafarm BV et Adriaan de Peijper v Winthrop BV; and 23 May 1978, in case C-102/77, Hoffmann-La Roche et Co. AG v Centrafarm Vertriebsgesellschaft Pharmazeutischer Erzeugnisse MBH. On Trademarks the legality was confirmed also under the Trademark Directive on 16 July 1998, in case C-355/96, Silhouette International Schmied GmbH v Hartlauer Handelgesellschaft GmbH.)).
Independent distribution of medicines within the Single Market is based on Articles 34 and 35 of the Treaty on the Functioning of the European Union (TFEU), subject to the derogations regarding the protection of human health and life and the protection of industrial and commercial property, provided by Article 36 of the TFEU.
Independent distribution of medicines in the EU’s Single Market is furthermore protected by the principle of exhaustion of intellectual property rights ((See Trademarks Directive (Directive 2015/2436/EU), and jurisprudence from the CJEU. For case law on patents; 29 February 1968, in Case C-24/67, Parke Davis and Co. v Probel (principle) and 31 October 1974, in case C-15/74, Centrafarm BV et Adriaan de Peijper v Sterling Drug Inc. For Trademarks; 13 July 1966, in joint cases C-56/64 and C-58/64, Établissement Consten S.à.R.L. and Grundig-Verkaufs-GmbH v Commission; 31 October 1974, in case C-16/74, Centrafarm BV et Adriaan de Peijper v Winthrop BV; and 23 May 1978, in case C-102/77, Hoffmann-La Roche et Co. AG v Centrafarm Vertriebsgesellschaft Pharmazeutischer Erzeugnisse MBH. On Trademark the legality was confirmed also under the Trademark Directive on 16 July 1998, in C-355/96, Silhouette International Schmied GmbH v Hartlauer Handelgesellschaft GmbH.)).
The exhaustion principle prevents right-owners from restricting further distribution of their products once they have placed these on a given EEA market themselves. That is because the IP owners have already extracted their ‘ownership profit’ with the first sale in the Single Market. This right cannot be used to obtain a double profit from IP.
ENTREPRENEURSHIP
Parallel importers and exporters are true entrepreneurs. Their contribution to innovation in the pharmaceutical sector lies in their ability to identify and procure products with relevant price differentials and thus foster competition and provide a service to patients by making these medicines more affordable. The Parallel distributors must meticulously research, calculate and meet the costs associated with purchasing, transport, warehousing, insurance, repackaging, quality assurance, regulatory compliance, distribution and marketing.
Regulatory framework of independent distribution
Parallel distribution is regulated under the Directive on Medicines for Human Use (Directive 2001/83/EC), the Falsified Medicines Directive (Directive 2011/62/EU), the Trade Marks Directive (Directive 2015/2436/EU), the Regulation for the authorisation and supervision of medicinal products for human and veterinary use and establishing a European Medicines Agency (Regulation (EC) No 726/2004), the Commission communication on the Community marketing authorisation procedures for medicinal products (Commission Communication 98/C 229/03) and national legislation.
Different requirement befit the two types of distributors, exporters and importers, in the market.
Exporters must possess:
- a wholesaler dealer license (Good Distribution Practice (GDP) license), and
- a Controlled Drug Authorisation, if they export controlled drugs.
Importers must possess:
- the GDP license,
- a Marketing Authorisation (allowing them to market a given medicine in a given market),
- a Manufacturing License (Good Manufacturing Practice (GMP) license) if they make changes to the label or outer package as demanded by national or local law, and
- a Controlled Drug Authorisation, if they import controlled drugs.
All authorisations and licenses are granted and controlled by the national competent authorities or by the European Medicines Agency.
Functioning of the sector
When a new medicine is created, its original creator gets a patent to protect the innovation. The duration of the patent protection depends on the applicable legislation – in the EEA, on EU-wide pharmaceutical legislation. The patent-holder of the medicine can prohibit other companies from manufacturing and selling their patent protected medicine. However, they cannot prohibit licenced wholesalers who have bought the product from the patent-holder from reselling the medicine in other countries within the EEA, where the patent-holder sells it at a higher price.
Prices of medicines vary for two main reasons: manufacturers maintain pricing strategies whose purpose is to maximize their profits, and the regulations defining the prices and reimbursements of medicines are set nationally, not on a common basis on EU level.
Independent distributors buy mostly patent-protected pharmaceuticals and under strictly regulated conditions move medicines from one market to the destination market. Some entrepreneurs in independent distribution export medicines, some import them into a destination market. Some actors in the sector undertake both export and import activities. These actions are called parallel exports and parallel imports. Trade in medicines flows in all directions in the Single Market, despite some countries having lower average medicine prices. This is because the price of an individual drug can be much lower in a traditionally high-priced market than in the average low-priced market. In fact, contrary to a common misbelief, more than 50% of all imported medicines originate from high-income countries.
Parallel imports are the only form of price competition during the validity of the patent protection of a medicine. In other words, parallel imports in medicines create competition on a sector where patents give the patent-holder a monopoly in every national market – and therefore almost an unlimited opportunity to define the price of the medicine, which can, in many cases, be extremely high. Price competition provided by parallel import is good for the European economy, good for health care systems and good for patients.
Safety
Parallel imports are original European supply sourced in another country of the EU/EEA and totally adapted to the destination market. These medicines are 100% safe, as parallel traders are subject to the same regulatory requirements as manufacturers of the branded or generic pharmaceuticals, and they have to undergo regular inspections by the competent authorities.
Savings
The objective of parallel trade is to offer Europeans original supply of medicines at a lower price. This creates sizable savings for public health insurance systems, pharmacies and, ultimately, patients. The savings stem from the price differences of manufacturers’ prices for the same medicine between countries of the EEA. There are direct and indirect savings.
Shortages
Although parallel trade is wrongfully blamed for provoking shortages or deteriorating the supply of medicines in European countries, there is little evidence to back these accusations. On the contrary, parallel trade has the ability to mitigate shortages by bringing medicinal products that are suffering from problems of supply in one country from another market in which there is a surplus.